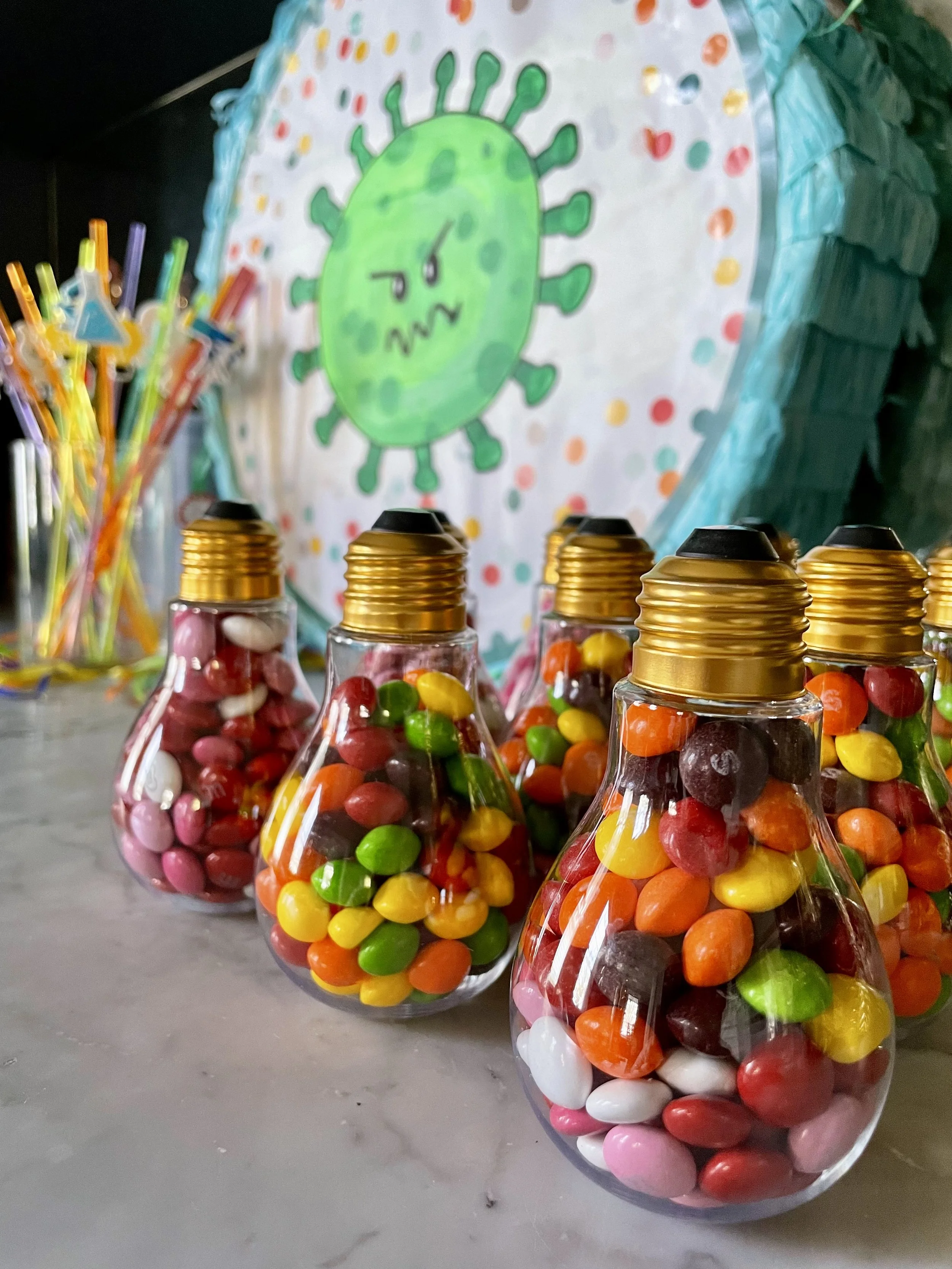Super Science Party!
Last year, my daughter had four party themes (Frozen, Gabby's Dollhouse, Unicorns, and Bluey, because why have one theme when you can have four?!). This year, after exploring every party option possible for her sixth birthday party—art, gymnastics, chocolate making, dinosaurs, museums, you name it—she narrowed it down to one: "super science"!
A lot of research and preparation went into planning this party. So much so that I decided to share it all in my inaugural blog post. Read on for all the party planning details, from the invitation to the experiments to the goody bags. I hope this post inspires other mini-scientists and helps save their party planners time!
The invitation…
Together, my daughter and I used Canva to design an invitation that hinted at the science fun to come: creating a chemical reaction to blow up balloons, building molecules with candy, mixing sparkly slime, crystal rock candy, and jello Petri dishes!
Paperless Post was used to send custom invitations and track responses.
The equipment…
On the big day, the mini scientists chose a letter patch to personalize their lab coats, colorful safety glasses, disposable gloves, a molecule pen, and science notebook before embarking on a series of experiments.
The experiments…
Experiment 1: Blow Up a Balloon!
This was probably the easiest and most fun experiment which elicited the most wows from the mini scientists!
Supplies: marble balloons, distilled white vinegar, empty bottles, a small funnel, baking soda
Directions:
Fill each bottle up halfway with the distilled white vinegar
Blow up the balloons to stretch them out a bit. Once deflated, insert the funnel into the balloon. Add 2 tbsp of baking soda to the balloon
Being careful to keep the baking soda in the balloon, tightly attach the end of the balloon to the lip of the bottle. Leave the top of the balloon hanging over the edge of the bottle until go time (see image below)
Lift the top of the balloon to pour the baking soda into the distilled white vinegar. Gently shake the bottle and set it down on a solid surface for the reaction to occur and watch the balloon inflate!
Simple science: The combination of the acid (vinegar) + base (baking soda) produces carbon dioxide gas, which inflates the balloon
Experiment 2: Let's Make Molecules!
The mini-scientists enjoyed eating their molecule creations as much as they enjoyed making them, maybe even more!
Supplies: Dots candy, toothpicks, molecule-building instruction cards
Directions: Use the toothpicks to connect the colored candy dots (elements) as shown by the black lines on the molecule reference cards
Note: Next time I might pre-allocate dots and toothpicks on individual plates to avoid a mass-grabbing situation!
Simple science: The candy dots represent the different elements (like Carbon, Hydrogen, and Oxygen) that molecules (like carbon dioxide, water, and methane) are made of. The toothpicks represent the chemical bonds that connect the elements to make the molecule
Experiment 3: Slime Time!
Slime time was the trickiest experiment that I wish I'd taken the time to test ahead of the party. The ratios of ingredients did not work as expected based on the instructions I’d found on Elmer’s website, perhaps because I subbed in Target glue for the pricier Elmer’s glue. Everyone still had fun mixing their choices of glitter glues and magical solutions though! I was glad it was an unexpectedly warm and sunny day that allowed us to conduct the stickiest experiment outside.
Supplies: 120 mL measuring cups with lids, stirrers, pipettes, washable school glue, Elmer’s glitter glue, Elmer’s Magical Liquid Slime Activator Solution, optional: glitter or food coloring add-ins
Directions: I suggest heading to Elmer’s website and searching for “slime recipe” to find instructions specific to the type of glue you are trying to make, given that the directions on my sign below came out very runny!
Simple Science: Slime is made when a polymer (glue) and gelling agent (eg, contact lens solution or Elmer’s Magical Liquid Slime Activator Solution) are mixed. Polymers are long strings of repeating molecules (or monomers). The gelling agent can stick to two strings of polymers and bind them together. As the gelling agent sticks to more and more strings of polymers, the solution becomes thicker and slimier - slime!
The treats…
We 'inoculated' red and green jello Petri dishes with gummy worms and sprinkles (the worm version could also work for a Halloween party, in a Petri dish or a cauldron).
The sprinkle versions looked so realistic that my husband was concerned that we were growing bacterial colonies in the fridge! Word of warning: wait until the jello is fully set and add the sprinkles at the last minute (vs the day before the party) or you'll risk them dissolving and forming puddles on the jello).
More candy came in the form of a Bad Bug Piñata! I made this by using white paint to cover the “happy birthday” words on this piñata and then painted on the main virus character from my book, Bad Bug Busters: Vaccines vs. Germs. The virus made a great target!
The party came to a close with ice cream cake and goody bags containing the cutest science stickers, straws, candy-filled plastic light bulbs, and hot pink crystal candy sticks.
All in all, the Super Science Party was a success and well worth the research and prep time!
To download free printable instruction cards for all of the experiments, subscribe to the newsletter…
Drop your comments or questions below!